Tiffany Lee, March 7, 2018 | View original publication
Physicists observe long-sought nanoscale phenomenon
Preparing the perfect nanoscale sandwich from oxygen-based ingredients was no picnic.
But with the assistance of two Nebraska physicists, an international team of researchers has finally managed it — ending a nearly 15-year quest to observe a phenomenon that could help power and miniaturize a future generation of electronics.
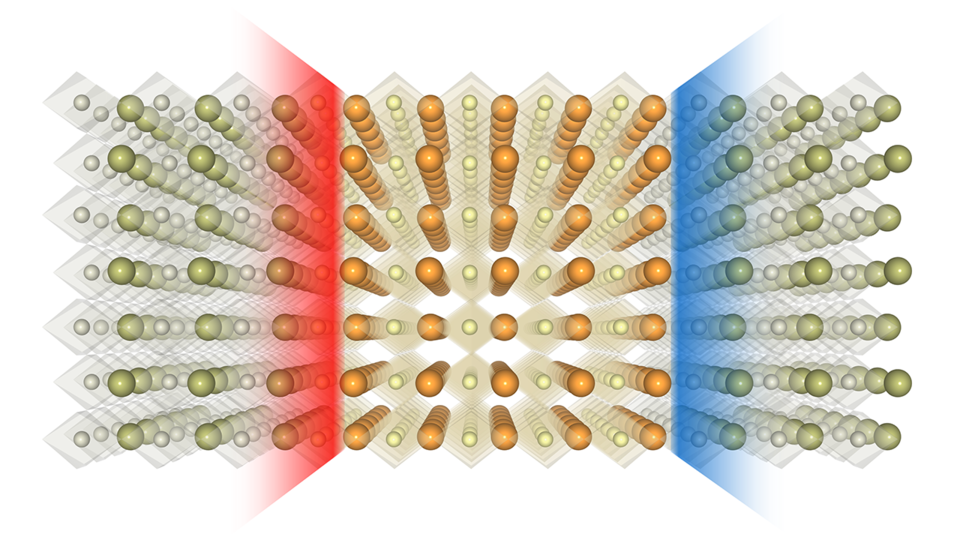
In 2004, researchers observed a gas of electrons coursing two-dimensionally through a nano-sandwich made from oxides: chemical compounds containing oxygen atoms. That demonstration of a 2-D electron gas signaled the promise of confining electric current to smaller spaces and, in turn, shrinking electronic components to smaller scales.
Yet the negatively charged electron has a counterpart – a positively charged “hole” it leaves behind when ejecting from its orbit around an atom. So physicists set out to create and observe a 2-D hole gas that likewise acts as a source of electric current.
As detailed in the journal Nature Materials, researchers from the University of Wisconsin-Madison and University of Nebraska-Lincoln led the way in accomplishing the long-sought feat. Doing so required several years of perfecting both the ingredients and preparation. Nebraska’s Evgeny Tsymbal and Tula Paudel informed the latter by running theory-grounded calculations and modeling through the university’s Holland Computing Center.
The recipe itself seemed simple enough. To produce a 2-D electron gas, researchers had previously stacked a positively charged oxide layer on a neutral base, finding that negatively charged electrons flocked downward to the nanoscopic space between the two. By adding a negatively charged layer atop the positive slice, then capping the nano-sandwich with another neutral layer, researchers had hoped to see positively charged holes mimic that behavior by migrating upward to form their own 2-D gas.
They resisted. Why? Oxygen atoms were abandoning their posts, and their positively charged vacancies — useless for producing an electric current — prevented the holes from moving on up.
“We looked at the different concentrations of the oxygen vacancies, the different positions of these defects, and how the behavior changes (as a result),” said Tsymbal, George Holmes University Professor of Physics and Astronomy.
The team found that it could get away with a few absentee oxygen atoms so long as those in the thick of the action managed to hold steady.
“Positioning is important,” said Paudel, a research assistant professor who performed most of the calculations. “You don’t want the oxygen vacancies near the region where you’re supposed to have a two-dimensional hole gas.”
Those insights, combined with precise specifications for the thickness of each slice in the nano-sandwich, guided experiments taking place at Wisconsin. By building those slices atom by atom — easier done with oxides than many other classes of materials — and fabricating the material in a pressurized, oxygen-rich environment that minimized vacancies, the Wisconsin researchers succeeded in producing and characterizing the 2-D hole gas.
Watch this space
For decades, engineers have fabricated the majority of electronic components from semiconducting materials such as silicon, the industry’s workhorse.
“The problem is that we are approaching fundamental limits,” said Tsymbal, director of Nebraska’s Materials Research Science and Engineering Center. “At some point (soon), we’ll approach certain limits beyond which we cannot continue (following) the semiconductor road map in the way that we did before. So we need to conceptually change the way our devices operate.”
One of those limits is space. The race to cram more functionality — memory, for instance — into tinier and tinier devices has left engineers looking toward oxides and other materials that, when combined, can squeeze conductivity into the tightest of confines. The new study took advantage of an oxide called strontium titanate — what Tsymbal described as the “silicon of oxide electronics” — to achieve this.
“The advantage here is that the confinement — the thickness of this two-dimensional electron or hole gas — is much smaller compared to what you have in semiconductors,” Tsymbal said. “Instead of, for example, tens of nanometers, we can confine it to one nanometer. So, in principle, we can make the devices much smaller as compared to those in semiconductor electronics.”
Though strontium titanate and its oxide brethren don’t generally exhibit magnetism on their own, they sometimes do when combined. They even show the potential for superconductivity — electric current that flows without any resistance — and other properties appealing to electric and computer engineers.
As theoreticians, Tsymbal and Paudel are interested in the phenomena that could emerge from 2-D electron and hole gases flowing in parallel through the same material. Among them: the pairing of electrons and holes into particle-like excitons that behave differently as a large collective than they do alone.
“Oxides boasting these complementary 2-D gases might now begin serving as nanoscopic laboratories in which to create and study new physics,” Paudel said.
How those phenomena might eventually be applied remains an open question, Tsymbal said, but one well worth exploring.
“When researchers started working on semiconductors more than 60 years ago, nobody knew that they would become central to modern technology,” Tsymbal said. “At this point, oxide electronics are on the level of fundamental research, so it’s hard to predict where they will go.
“But you can control oxide interfaces with extreme precision. Once you have that, you can do something similar to what semiconductors achieve — but maybe also something else.”
The Wisconsin and Nebraska researchers authored the Nature Materials study with colleagues from Ohio State University, Sungkyunkwan University (South Korea), Pohang University of Science and Technology (South Korea) and Argonne National Laboratory.
The team received support from the National Science Foundation and the U.S. Department of Energy’s Office of Science.