Scott Schrage, August 25, 2023
Study IDs secret of stealthy invader essential to ruinous rice disease
FINDINGS COULD INFORM TREATMENT OF PATHOGENS COMMON IN PLANTS, PEOPLE
The virulence of a rice-wrecking fungus — and deployment of ninja-like proteins that help it escape detection by muffling an immune system’s alarm bells — relies on genetic decoding quirks that could prove central to stopping it, says research from the University of Nebraska–Lincoln.
A Nebraska team helmed by Richard Wilson hopes that identifying an essential but formerly unknown stage in the fungal takeover of rice cells can accelerate the treatment or prevention of rice blast disease, which ruins up to 30% of global yields each year.
“The response I’ve gotten from people in my field is that they’re very excited, because nobody’s been able to get a handle on this,” said Wilson, professor of plant pathology at Nebraska.

Most cellular machines, or proteins, are secreted in the same way: After being constructed and folded into their near-final forms at the endoplasmic reticulum, they move on to the Golgi body, which packages and forwards them along to their ultimate destinations. But certain proteins will bypass the Golgi body in favor of unconventional, poorly understood pathways. Wilson’s team has now shown that one of those unconventional pathways involves modifying not the secreted protein itself, but the genetic code of a molecule that aids in its construction.
Known as transfer RNA, or tRNA, that molecule hauls around amino acids — the building blocks of every protein — in search of a blueprint that calls for its particular cargo. Those blueprints exist as three-letter codes, or codons, carried by the fittingly named messenger RNA. When a tRNA comes across and decodes an mRNA whose codon matches its own three-letter combination, it unloads its corresponding amino acid, adding it to a string of others that ultimately yield a finished protein.
Before giving up their precious cargo, though, some tRNAs undergo chemical makeovers. One especially notable modification? The addition of sulfur to the tRNA’s third letter, or nucleotide — specifically when that letter is U, the nucleotide known as uridine. Though that sulfur addition has been conserved and observed in a wide range of organisms, from yeast to mice to humans, researchers have yet to pin down all its functions.
Wilson and his colleagues decided to play an educated hunch: that the modification of the tRNA’s uridine might prove important to the growth of Magnaporthe oryzae, the fungal species that causes rice blast disease. To test its importance, the researchers resorted to the tried-and-true method of removing the genes responsible for the modification, then looking for any differences between that mutant fungus and its original counterpart.
The team discovered even more than it bargained for. Gone were some of the ninja-like proteins, or effectors, that are secreted via the unconventional pathway before infiltrating the cytoplasm of rice cells to mitigate their innate immune responses. And when the team deposited spores from the mutant M. oryzae on rice seedlings, the effector-less fungus mustered only minuscule lesions on their leaves — lesions far smaller than those managed by the untouched, virulent fungus.
That sulfur-modified tRNA could assist the search for disease-enabling effectors in M. oryzae and a rash of other pathogens, Wilson said. In the case of M. oryzae, tRNAs were consistently matching with mRNA codons ending in AA — adenine in the second and third positions of the codon. Yet the team knew that other tRNAs could also match with synonymous codons that instead ended in AG, unloading the very same amino acid when they did — and without the fuss of adding sulfur beforehand. Which left a question: Why, exactly, did M. oryzae prefer the AA-ending codons to their AG-ending peers?
Another experiment would help solve the mystery. Swapping out the AA-ending codons for AG, the team found, did lead the M. oryzae to resume its production of the virulent effector proteins. Unfortunately for the fungus, it began cranking out so many effectors that they effectively disrupted their own stealth operation and ultimately failed to facilitate infection, the result of far too many cooks in a nanoscopic kitchen. The AA-ending codons, it turned out, were not only enabling but also regulating the production of the effectors. It was clear: The stealthy proteins depended on both the sulfur modification and one specific, calibrating type of codon being targeted by it. If either was missing, the whole gambit fell apart.
Because mRNA extracts its blueprints directly from the source code known as DNA, analyzing the latter can allow researchers to discern the presence and prevalence of codons in the former. Knowing just how heavily M. oryzae depends on AA-ending codons to churn out the effectors that invade a rice cell’s cytoplasm, Wilson and his colleagues went looking for signs of them in relevant genes. The team was not disappointed: In one case study, more than 90% of the AG- and AA-ending codons for a cytoplasmic effector fell into the latter category.
Researchers could easily seek out the same telltale disparity for the sake of tracking down more effectors — whether in fungus or other pathogenic organisms — that perpetuate sneak attacks, Wilson said.
“One of the goals of plant pathology is to identify new effectors and figure out their functions, which are often to inhibit the function of some plant protein or evade detection,” he said. “And then, if you find that target in the plant, you can make changes in the plant to make them more resistant. So finding effectors is really a search for durable plant disease resistance.
“I think, in the near term, we’ll use this to leverage more of an understanding of this unconventional secretion pathway. It’s the only pathway known for the blast fungus to get effectors into a plant cell, so if you can inhibit it in some way, then it would really be detrimental to the fungus in terms of being able to cause disease.”
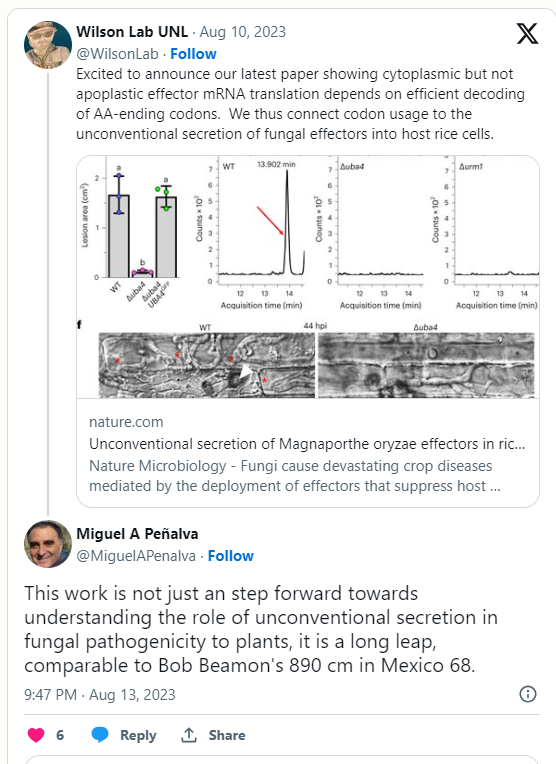
An acclaimed cellular biologist not involved in the study compared the team’s achievement to that of Bob Beamon, who sailed nearly 2 feet past the then-world record in the long jump at the 1968 Summer Olympics.
The unconventional effector pathway studied by the team encompasses more than just the kingdoms of fungi and plants, Wilson said. Parasitic protists that drive many human-contracted diseases, including malaria, secrete immune-snuffing proteins in much the same way as M. oryzae. Some cancer cells also use the pathway. Ideally, Wilson said the team’s study could inform efforts to both identify new effectors and understand why they emerged as weapons of choice against human cells, too.
“So there are many ways,” he said, “in which this work could enlighten a whole host of things.”
The researchers reported their findings in the journal Nature Microbiology. Richards authored the study with Gang Li and Nawaraj Dulal, both with Nebraska’s Department of Plant Pathology, alongside Ziwen Gong of the Chinese Academy of Agricultural Sciences. The team received support in part from the National Science Foundation.